Everyone knows about FAT: we all care about it and everyone worries about it. Fat is among the most popular topics in the urbanized world, perhaps slightly less of a concern to the rural population and in some societies, an asset to some few.
Our conversations about FAT are limited, most of the time, to “White” Adipose Tissue, referred to in the medical literature as WAT; most of us know nothing about brown fat – our Brown Adipose Tissue, jocularly nicknamed BAT. [1]
WAT
The infinitely expandable energy storage layer of WAT fills out the areas between our bony protuberances, providing the “curvaceous” outline in which healthy young women take pride and without which we don’t “look well”, but it is distressingly liable to expansion, especially in those were subject to chronic stress and its most frequent sequel, intracellular hypothyroidism. [10]
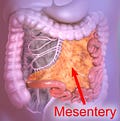
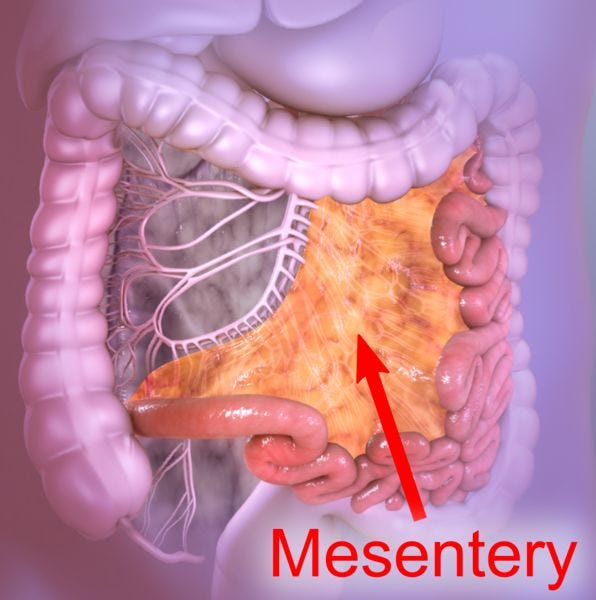
WAT is also found in many other parts (listed below); the most spectacular of which is the mesentery (see graphic 1, below).
Graphic 1, from Wikipedia: the article is at https://en.wikipedia.org/wiki/Mesentery.
As well as our subcutaneous fat layer, we carry WAT in a lot of places:
(1) in the “mesentery”, a double-layered, transparent sheet of peritoneum that looks like cellophane, attached to the midline, from which the small bowel hangs (Graphic 1).
A similar sheet hangs off the stomach, to support the middle part of the large bowel: it also contains some WAT; but less than the small intestinal mesentery.
(2) Inside the pericardium (the sac which holds the heart).
(3) Around the outside of the peritoneum (the lining of the abdominal cavity).
About the only places where there isn’t any white fat are inside the skull, in the gums, between the pleura (the lining of the chest cavity) and the ribs, the genitalia (testicles and ovaries) and the nail beds of the fingers and toes.
BAT
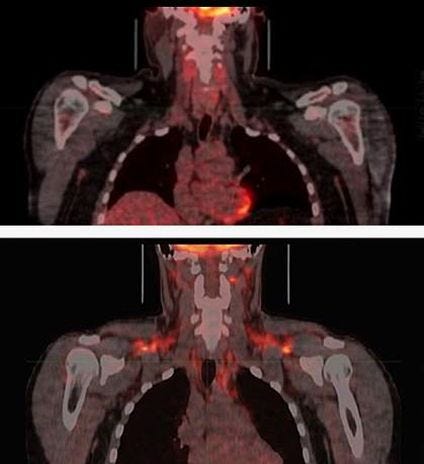
The BAT, a completely different, fixed-volume, energy-burning organ, is situated discreetly beside the spine, in the shoulders, the base of the neck and the “mediastinum”, where the heart, the esophagus and our “breathing pipes” lie hidden, deep within the chest.
Graphic 2, from “ice bath science”, an excellent post on brown fat by Thomas P. Seager, PhD; read Dr. Seagear’s paper, at https://www.morozkoforge.com/post/ice-bath-science
The difference between WAT and BAT:
WAT
WAT has a sparse blood and nerve supply and is composed of large, unilocular cells, containing a single large lipid droplet and only few small mitochondria. The massive increase in WAT, seen in obesity, is due to enlargement of the individual fat cells to many times their original size, by deposition of unused oil and storage fats, in the form of BCAAs (branched-chain amino acids) from what we eat.
BAT
In contrast, BAT adipocytes are small, multilocular cells, containing multiple small lipid droplets and numerous mitochondria with high iron and cytochrome content. They are densely innervated by the sympathetic nervous system and are highly vascularized. They do not import unused fatty acids from the diet and do not enlarge in the way that WAT cells do.
Under special circumstances, BAT can enlarge itself by producing new BAT cells, from its progenitor, “stem”, cells. Thus the volume of BAT can be increased , but this is a true growth of the organ, as distinct from enlargement of the individual cells, as seen in WAT.
WAT contains a few stem cells, which can be converted to BAT-type cells under special circumstances (prolonged cold weather, or stimulation by long-term therapy with T3 hormone). When this conversion occurs, the affected WAT cells become “brownish” and have been referred to as “beige cells”: they produce heat in the same fashion as brown fat cells do, but they are not as efficient.
Anyway, everyone knows about white fat, so let’s talk about brown fat, first.
FAT connections, function and value
White Fat (“WAT”)
White Adipose Tissue is a storage bin.
When we eat more calories than we need, glucose and “fatty acids” called BCAAs (Branched-Chain Amino Acids) from our food enter the lipocytes, as white fat cells are called, and are stored in the form of triglycerides.
The lipocytes readily enlarge to many times their original size, accepting as much glucose and fatty acids as happens to be available. Once stored, the fats and oils simply sit in the lipocytes, waiting for the day when food intake dips below calorie usage.
White fat has no other purpose under normal circumstances, but under conditions of prolonged cold weather, its few mitochondria can be recruited to produce heat: when this happens, there is a slight, brownish colour change and the modified cells are referred to as “beige fat”.
Brown Fat (“BAT”)
Brown Adipose Tissue is an active organ, whose cells contain a greater-than-usual collection of energy-producing mitochondria. We always thought that the BAT, which shrinks as we get older, was not expandable; but recently, it has been demonstrated that long-term therapy with T3, or prolonged cold weather, can signal BAT’s progenitor cells (stem cells) to multiply and develop into new brown fat cells. [2,4]
Brown fat does not store energy: it produces it. When it is cold, brown fat cells absorb glucose, triglycerides and “non-HDL-cholesterol”, plus “BCAA” (the “branched” amino acids, Leucine, Isoleucine and Valine), from the blood. [3] Their mitochondria “burn” these molecules by a chemical process (“thermogenesis”) which produces heat, so that our temperature stays normal.
Thermogenesis is triggered by release of Noradrenaline into BAT, by the sympathetic nervous system. However active burning doesn’t start unless there is an adequate supply of functioning thyroid hormone, T3*. So providing that there is enough T3 in the BAT cells **, excess food is used to keep the body temperature normal, minimizing the liability to over-stuffing the white fat with BCCA and glucose.
Also, burning BCCA’s and glucose reduces the glucose and cholesterol in the system, Our BAT keeps our A1C and cholesterol normal and minimizes our liability to diabetes, hypercholesterolemia and atherosclerosis, thus protecting us from heart attacks, strokes, large vein blood clots, etc. [5, 5a]
*T3 doesn’t trigger thermogenesis without noradrenaline and noradrenaline doesn’t start thermogenesis if no T3 is present.
**Recently, it has been reported that T2 (Diiodothyronine) will also support thermogenesis: see “CAVEAT”, below.
Babies are born with quite a lot of brown fat, so they are good at staying warm. However humans have a tendency to reduce the amount of brown fat in the body as we get older and for some reason, we don’t increase its volume much, as we grow: some people have very little, or no, BAT. [4]
Caveats:
– It is tempting to think that older folks feel the cold more (they do!) because of a reduction in their brown fat volume. However the real reason is that with age, the production of thyroid hormone falls, so that humans over 70 years of age tend to be hypothyroid: without sufficient thyroid 3 hormone, brown fat lies dormant.
– It may be, that the shrinkage of the brown fat occurs because of reduction of availability of thyroid hormone with aging: this is an unrecognized problem in older folks, because mainstream medicine overestimates the importance of TSH (thyroid stimulating hormone), estimates a much-too-wide “normal” TSH range and under–diagnoses hypothyroidism: in 2005, Wartofsky and Dickey showed that more than 95% of normal individuals have TSH levels below 2.5 mU/liter; but few doctors seem to have heard about it.
– Some investigators report that T4 is converted to T3 by DEIODINASE 2 in BAT: if so, BAT must be immune to Intracellular Hypothyroidism – this idea is a potential game-changer, vis-a-vis the effect of stress on BAT. I will have to report on that aspect, later!
Food
Turmeric (curcumin), resveratrol, green tea and capsaicin (pepper) may activate brown fat, initiating thermogenesis; but this is a mild effect.
Caffeine, ginseng, quercetin and various other extracts have been shown to activate brown fat in rats, but their thermogenic effect in humans is negligible.
Exercise
Sufficient exercise may succeed in oxidizing some white fat to “beige”, but it does not activate the brown fat and does not result in an increased volume of brown fat.
“Cryotherapy”
Cold showers and ice baths have been proposed [1], to activate the brown fat; but the duration of any temperature drop achieved is short. Frequent walks in winter weather may help, but that is generally futile, in terms of producing weight loss.
STATIN DRUGS INHIBIT BROWN FAT ACTIVITY
An interesting report [2], suggests that commonly prescribed cholesterol-lowering statin drugs reduce the volume of BAT in adults. The team’s in vitro work and in vivo studies in mice showed that statins reduced brown fat function and the “browning” of white fat cells into beneficial BAT.
A study of imaging data from thousands of humans also suggested that statin use was inversely correlated with active BAT.
Brown Fat thermogenesis reduces blood sugar and cholesterol
Brown and beige adipocytes enhance “lipolysis”, the breakdown of triglyceride-rich lipoproteins in the blood, to supply them with fatty acids for oxidation.
Activation of thermogenic adipocytes thus lowers circulating triglycerides and non-HDL-cholesterol and increases HDL-cholesterol: the combined effect is protection from atherosclerosis and the heart attacks and strokes which it can cause. [8]
In an interesting study, entitled “Role of thermogenic adipose tissue in lipid metabolism and atherosclerotic cardiovascular disease: lessons from studies in mice and humans”, published in Cardiovascular Research, Volume 119, Issue 4, April 2023, Pages 905–918, https://doi.org/10.1093/cvr/cvac131, Zhixiong Ying et al. concluded that activated Brown adipocytes in BAT and beige adipocytes in WAT not only dissipate nutritional energy as heat; they absorb triglycerides and non-HDL-cholesterol from the blood, thus protecting the body from atherosclerosis. [8]
Triiodothyronine, “T3”, gets your BAT swingin’!
In an excellent, but rather a long and difficult to read, article written in 2022, entitled “T3 promotes brown fat hyperplasia via thyroid hormone receptor alpha–mediated adipocyte progenitor cell proliferation”, Shengnan Liu et al. remind us that enhancing the activity of BAT is a promising strategy for prevention and treatment of obesity and its associated metabolic diseases.
Utilizing novel experiments in rats, they showed that T3 increases the thermogenic capacity of BAT, via proliferation of adipocyte progenitor cells.
According to Liu et al., 50 g of active BAT can produce a 20% daily increase in energy expenditure in humans, so any increase in active BAT volume is likely to result in a significant increase in consumption of glucose and BCCA, resulting in weight loss.
In hypothyroid patients, brown fat doesn’t function well and there is little thermogenesis in response to cold: that’s why hypothyroid subjects are so often heard to say “I always feel cold!”. Treatment of hypothyroidism with thyroid hormone returns the brown fat to normal activity and thermogenesis is restored. [6]
Brown Fat protects health
Between June 2009 and March 2018, Tobias Becher, M.D., and Paul Cohen, M.D., Ph.D., of the Laboratory of Molecular Metabolism at The Rockefeller University in New York, collaborating with colleagues at the Memorial Sloan Kettering (MSK) Cancer Center, studied 134,529 PET/Computed Tomography reports from 52,487 patients treated at MSK, documenting the presence or absence of brown fat and matching each patient’s scans with their other clinical findings. [5]
They found that the odds of having cardiovascular and metabolic disorders were lower when patients had brown fat.
For example, patients with brown fat had:
56% lower odds of having type 2 diabetes,
14% lower odds of having high cholesterol and triglycerides,
32% lower odds of coronary artery disease, and 15% lower odds of high blood pressure.
Brown fat was even protective in the obese: obese patients who had brown fat had lower odds of cardiometabolic conditions, especially type 2 diabetes, coronary artery disease and congestive heart failure.
Prime Factors, From the point of view of a metabolic medicine MD
– Children generally, are “skinny”, in spite of an active appetite.
– Humans reduce the percentage of brown fat as they age.
– Production of DHEA and neurosteroid hormones (especially, testosterone) is reduced by 1% per annum, beginning at age 26.
– Many, who are slender in youth, begin to gain weight and to exhibit other hypothyroid symptoms such as hair loss, between age 26 and 40.
– Most of those who do not fatten in the 3rd or 4th decade, begin to gain weight in the 5th or 6th, as a result of hormonal changes (particularly, females tend to gain weight in the menopause).
– Obesity is endemic in modern, high-stress societies (PLEASE look this one up).
– There is a natural reduction of thyroid hormone production and activity with age, reflected in a continuous increase in thyroid stimulating hormone (TSH) levels and in addition to this, stress-related intracellular hypothyroidism is endemic, particularly in city-dwellers.
– Hypothyroidism is associated with downregulation of brown fat activity, as evidenced by low body temperature in hypothyroidism.
– Hypothyroidism, particularly intracellular hypothyroidism, is associated with lethargy, reduced cognition, low motivation levels, depression and weight gain.
– Hormonal imbalance, notably DHEA deficiency, is frequently associated with intracellular hypothyroidism. The patients, predominantly female, present in their 20s or early 30s, with typical hypothyroid symptoms: hair loss, brittle fingernails, weight gain, reduced libido, anxiety and occasionally, cognitive loss. The serum DHEA levels are usually < 5 µmol/litre.
Supplementation with oral DHEAS reliably relieves the hypothyroid symptoms (unpublished data, from 600 cases), suggesting that DHEA improves intracellular T3 levels.
Therefore, I postulate as follows:
(A) It is likely that obesity is due to downregulation of BAT function, by progressive loss of functioning triiodothyronine, whether due to loss of DHEA production (1% per annum, from age 26), reduced thyroid gland function from other causes, or to Intracellular Hypothyroidism. (There remains the question as to why some people are devoid, or almost devoid, of brown fat, [8] but I will defer that for now).
(B) Rather than awaiting the development of obesity, diabetes and atherosclerosis (or, God forbid, a heart attack!), a better idea would be to institute preventive care, with proactive surveillance of renal, hepatic, neurosteroid and thyroid function, beginning in adolescence. [9]
(C) Aberrations of hormonal balance, observed at whatever age, would provide early diagnosis of neurosteroid deficiency and/or intracellular hypothyroidism (click here), permitting early correction of the factors which lead to obesity.
(D) Neurosteroid imbalance is easily and safely corrected with bioidentical hormone restoration therapy, beginning at age 26 with DHEA (click here).
Intracellular Hypothyroidism is treated (also easily and safely) with slow-release oral triiodothyronine.[10]
CAVEAT: diiodothyronine (T2) is not a substitute for T3!
Arturo Hernandez, PhD, published an article entitled “3, 5–Diiodo–L–thyronine (T2) in dietary supplements: what are the physiological effects?” in the January 2015 issue of the journal, “Endocrinology”, 156(1): 5–7, doi: 10.1210/en.2014-1933, PMCID: PMC4272398, PMID: 25526549. [
In his excellent article, Dr. Hernandez advises us that “a TH metabolite, 3,5-diiodo-L-thyronine (T2), has found its way into the formulations of a number of dietary supplements that claim to decrease body fat. They are available “over the counter,” and a quick internet search reveals that they are offered for sale using marketing verbiage such as “fat burner,” “appetite suppressant and fast fat annihilator,” “metabolic accelerator,” etc.”
In my opinion, the potential side effects of the massive doses (up to 80 times the dose, by weight) of T2 which might mimic the action of T3, are such as to categorically negate the use of such supplements: a better idea would be to discuss the general subject with a metabolic medicine professional. [11]
REFERENCES
[1] Ice Bath Science: Facts, Myths, and Mysteries, by Thomas P Seager, PhD, Jun 19, 2022, 17 min read, updated: Jan 20,
https://www.morozkoforge.com/post/ice-bath-science
[2] Inhibition of Mevalonate Pathway Prevents Adipocyte Browning in Mice and Men by Affecting Protein Prenylation (https://www.genengnews.com/news/cholesterol-lowering-drugs-inhibit-brown-fat-activity/”, by Christian Wolfrum, Ph.D., professor, of ETH Zurich (et al), was published in Genetic Engineering and Biotechnology News (Marianne Liebert Inc., publishers) on 12/24/18
[3] NIH Research Matters September 10, 2019 How brown fat improves metabolism
BCAA catabolism in brown fat controls energy homeostasis through SLC25A44. Yoneshiro T, Wang Q, Tajima K, Matsushita M, Maki H, Igarashi K, Dai Z, White PJ, McGarrah RW, Ilkayeva OR, Deleye Y, Oguri Y, Kuroda M, Ikeda K, Li H, Ueno A, Ohishi M, Ishikawa T, Kim K, Chen Y, Sponton CH, Pradhan RN, Majd H, Greiner VJ, Yoneshiro M, Brown Z, Chondronikola M, Takahashi H, Goto T, Kawada T, Sidossis L, Szoka FC, McManus MT, Saito M, Soga T, Kajimura S. Nature. 2019 Aug 21.
doi: 10.1038/s41586-019-1503-x. [Epub ahead of print]. PMID: 31435015. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6715529/
[4]Triiodothyronine (T3) promotes brown fat hyperplasia via thyroid hormone receptor α mediated adipocyte progenitor cell proliferation.byLiu S, Shen S, Yan Y, Sun C, Lu Z, Feng H, Ma Y, Tang Z, Yu J, Wu Y, Gereben B, Mohácsik P, Fekete C, Feng X, Yuan F, Guo F, Hu C, Shao M, Gao X, Zhao L, Li Y, Jiang J, Ying H. Nat Commun. 2022 Jun 13;13(1):3394. doi: 10.1038/s41467-022-31154-1. PMID: 35697700; PMCID: PMC9192766.
https://pubmed.ncbi.nlm.nih.gov/35697700/
[5] Becher T, Palanisamy S, Kramer DJ, Eljalby M, Marx SJ, Wibmer AG, Butler SD, Jiang CS, Vaughan R, Schöder H, Mark A, Cohen P. Brown adipose tissue is associated with cardiometabolic health. Nat Med. 2021 Jan;27(1):58-65. doi: 10.1038/s41591-020-1126-7. Epub 2021 Jan 4. PMID: 33398160; PMCID: PMC8461455.
[5a] CTSA Program-Supported Researchers May Turn Brown Fat into an Ally Against Obesity (updated: 03-23-2022)
https://ncats.nih.gov/pubs/features/ctsa-program-supported-researchers-may-turn-brown-fat-into-an-ally-against-obesity
[5b] Brown fat holds promise for addressing obesity and a host of related ills, by Marcus A. Banks, October 13, 2022, 119 (43) e2216435119, https://doi.org/10.1073/pnas.2216435119
[6] Resolution of Hypothyroidism Restores Cold-Induced Thermogenesis in Humans, by Claudia Irene Maushart, Rahel Loeliger, Gani Gashi, Mirjam Christ-Crain and
Matthias Johannes Betz Published Online:9 Apr 2019 https://doi.org/10.1089/thy.2018.0436
[7] Autophagy. 2019; 15(1): 131–150. Thyroid hormone (T3) stimulates brown adipose tissue activation via mitochondrial biogenesis and MTOR-mediated mitophagy, by Winifred W. Yau, Brijesh K. Singh, Ronny Lesmana, Jin Zhou,a Rohit A. Sinha, Kiraely A. Wong, Yajun Wu, Boon-Huat Bay, Shigeki Sugii, Lei Sun, and Paul M. YenPublished online 2018 Sep 13. doi: 10.1080/15548627.2018.1511263, PMCID: PMC6287687, PMID: 30209975
[8] Role of thermogenic adipose tissue in lipid metabolism and atherosclerotic cardiovascular disease: lessons from studies in mice and humans, by Zhixiong Ying, Naomi Tramper, Enchen Zhou, Mariëtte R Boon, Patrick C N Rensen, Sander Kooijman, in Cardiovascular Research, Volume 119, Issue 4, April 2023, Pages 905–918, https://doi.org/10.1093/cvr/cvac131
[9] Patient Surveillance in Family Practice 2022 12 09, by G. A. Harry, published via research gate, December 2022, DOI: 10.13140/RG.2.2.31292.82568, https://www.researchgate.net/publication/366187205_Patient_Surveillance_in_Family_Practice_2022_12_09
[10] Intracellular Hypothyroidism, by G. A. Harry, published via research gate, May 2023. https://www.researchgate.net/publication/370772568_INTRACELLULAR_HYPOTHYROIDISM
[11] 3, 5–Diiodo–L–thyronine (T2) in dietary supplements: what are the physiological effects?, by Arturo Hernandez, PhD: “Endocrinology”, 156(1): 5–7, doi: 10.1210/en.2014-1933, PMCID: PMC4272398, PMID: 25526549.